CALCULATIONS AND COMPARISONS OF SYSTEM CONDUCTANCES AND EFFECTIVE SPEEDS FOR TRANSITIONAL FLOW FOR VARIOUS PROCESS GASES
SYSTEM DESIGN V4
SECTION 1 – Design Overview
From the previous Chapter 3 posts, the calculations for molecular flow for system iterations V2, V3, and V4 for several major gas loads that would be encountered in future experiments were covered. The current design iteration is V4, which all following calculations will be based off of. Now that molecular flow numbers for total pipeline conductance and effective speed of the system have been established, the next phase can be calculated – transitional flow. Where molecular flow deals with the high vacuum regime and below (generally between 10^-4 Torr and lower), transitional flow deals with the low vacuum area between very rough pumping and the start of molecular flow. This is somewhat of a grey area, but can generally be thought of in the range roughly between 10^-1 Torr and 10^-3 Torr. This area of operation is particularly interesting for a variety of devices, including the fusor, as well as other higher-pressure process operations. For the following calculations, a pressure of 10^-2 was used.
As with molecular flow, transitional flow is calculated for each of the major system gases. The PDFs for the calculations are found below as follows:
2.75″ Conflat High Vacuum System – Design Iteration 4 – Transitional Flow – AIR
2.75″ Conflat High Vacuum System – Design Iteration 4 – Transitional Flow – ARGON
2.75″ Conflat High Vacuum System – Design Iteration 4 – Transitional Flow – DEUTERIUM
2.75″ Conflat High Vacuum System – Design Iteration 4 – Transitional Flow – WATER VAPOR
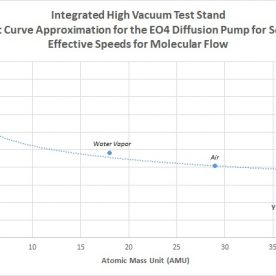
The first number to establish for the calculations can be seen in Section 1.) Diffusion Pump of the PDFs. Since the diffusion pump model is known and the data sheet can be accessed, the maximum speed of the pump can be determined, which is needed for the end calculations. Note that for molecular flow, for air as an example, the maximum speed of the pump is 600 L/s. This however is not the case for the entire pressure range of the diffusion pump. Diffusion pumps follow a certain speed vs. pressure curve, where the speed rises from a low value after the critical backing pressure, up to its maximum at some value in the high vacuum regime, where pump speed remains constant afterwards. Since we are looking to operate at 10^-2 Torr, this point on the curve must be found and approximated for the speed of the pump at this pressure. From the datasheet, this value roughly correlates to about 100 L/s. This becomes the maximum speed of the diffusion pump for calculating transitional flow for 10^-2 Torr. It can be seen that this much lower number will result in a larger discrepancy between the total pipeline conductance and the effective speed, whereas this difference is much smaller in molecular flow due to the much higher maximum speed of the pump.
The following sections from the PDF, Sections 2-7, deals with the calculations for transitional flow for each of the components in the pipeline. Note that molecular flow conductance is needed for these calculations. Also needed is the diameter of the component, as well as the mean free path. For 10^-2 Torr, the mean free path equates to about 0.5 cm. Due to the physics of the flow of gases between transitional and molecular flows, transitional flow conductances and speeds will be higher. This is also a good benefit for allowing for higher gas loads in the system at this pressure. The total conductances and effective speeds are calculated the same way as in molecular flow. Below are the resulting numbers of conductances and effective speeds for the various gases for the system V4 design:
AIR:
- Conductance – 18.621 L/s
- Effective Speed – 15.698 L/s
ARGON:
- Conductance – 15.881 L/s
- Effective Speed – 13.705 L/s
DEUTERIUM:
- Conductance – 73.204 L/s
- Effective Speed – 42.265 L/s
WATER VAPOR:
- Conductance – 23.610 L/s
- Effective Speed – 19.100 L/s
If you compare these numbers from the numbers for V4 for molecular flow, you will find that conductance and effective speed are higher for air, argon, and water vapor. The difference between conductance and effective speed is also larger for transitional flow as well, due to the lower maximum pumping speed at this increased pressure. While the conductance is much higher for deuterium between transitional and molecular flow, the effective speed turns out to be lower, based on the following calculation decisions from available data. For molecular flow, the max speed for deuterium is known at 800 L/s as opposed to 600 L/s for air and other similar gases. However, on the speed vs. pressure chart in the datasheet, only data was presented for air. To design for a worse case scenario, it was decided to also use the speed of 100 L/s for calculating deuterium at 10^-2 Torr, which is the same as the speed actually given from the datasheet for air. In reality this number will be higher for equivalent pressures between molecular and transitional flows, but since the exact curve is unknown, calculation for deuterium is based on a worse case basis to establish a lower bound for this number. Therefore, it is very reasonable to expect that the effective speed for deuterium in reality will be much higher than for molecular flow for the given pressure, since the maximum pumping speed for the pump for hydrogen will be higher at 10^-2 Torr than it is for air. In this regard, the total conductance and effective effective speed for water vapor should also be a bit higher than calculated, while the total conductance and effective speed for argon should be lower in reality
While argon remains on the low side, presently experiments with argon at these vacuum levels in the transitional flow regime are not planned. Air is included in all of these calculations because it is a gas that is most often referred to for experimental numbers and measurements in literature, as well as nitrogen, which provides a good comparison baseline. Now that the numbers for transitional flow have been established, the remaining parameters of the system can be calculated, including ultimate pumpdown pressure, outgassing loads, pumpdown times, and maximum loads for a given gas at a given pressure.